


Best known for our strong legacy in medical applications for powering ventilators and infusion pumps, SL Power is here to support the shortage of medical equipment we are experiencing worldwide. Power supplies for medical and healthcare equipment have to meet not only the functional parameters of your clinical application but also safety approvals such as IEC 60601-1-2 and 4th edition standard. This standard defines the basic and essential performance for medical equipment in regard to emissions and immunity to electromagnetic disturbances that help power medical equipment to include home use healthcare.
We have built a portfolio of standard medical grade power products that are TREPP optimally designed and outperform the most state of the art products within their class ensuring seamless integration with the end equipment. Our goal is to reduce the time to market, which saves on development cost and opportunity loss.
During this COVID-19 emergency, we are seeing a shortage in medical equipment and to combat this, we are staying true to our mission of saving and sustaining lives by proactively allocating resources to support the global need.

TREPP is not only an acronym representing the five performance dimensions of power conversion but a standard that SL Power’s engineers strictly adhere to. Optimizing TREPP is a primary consideration throughout the entire product development cycle and a driving force that guides engineering decisions.
We also have the processes and experience needed by medical equipment makers. Our proprietary customer-centric product development process identifies and mitigates design, engineering, manufacturing, and sourcing risk through industry standard (DFMEA, PFMEA, ISO 14971) and proprietary tools.
In-house evaluation and testing laboratories minimize expensive certification costs, ensure rapid development cycles and significantly improve the operational reliability of our power supplies.
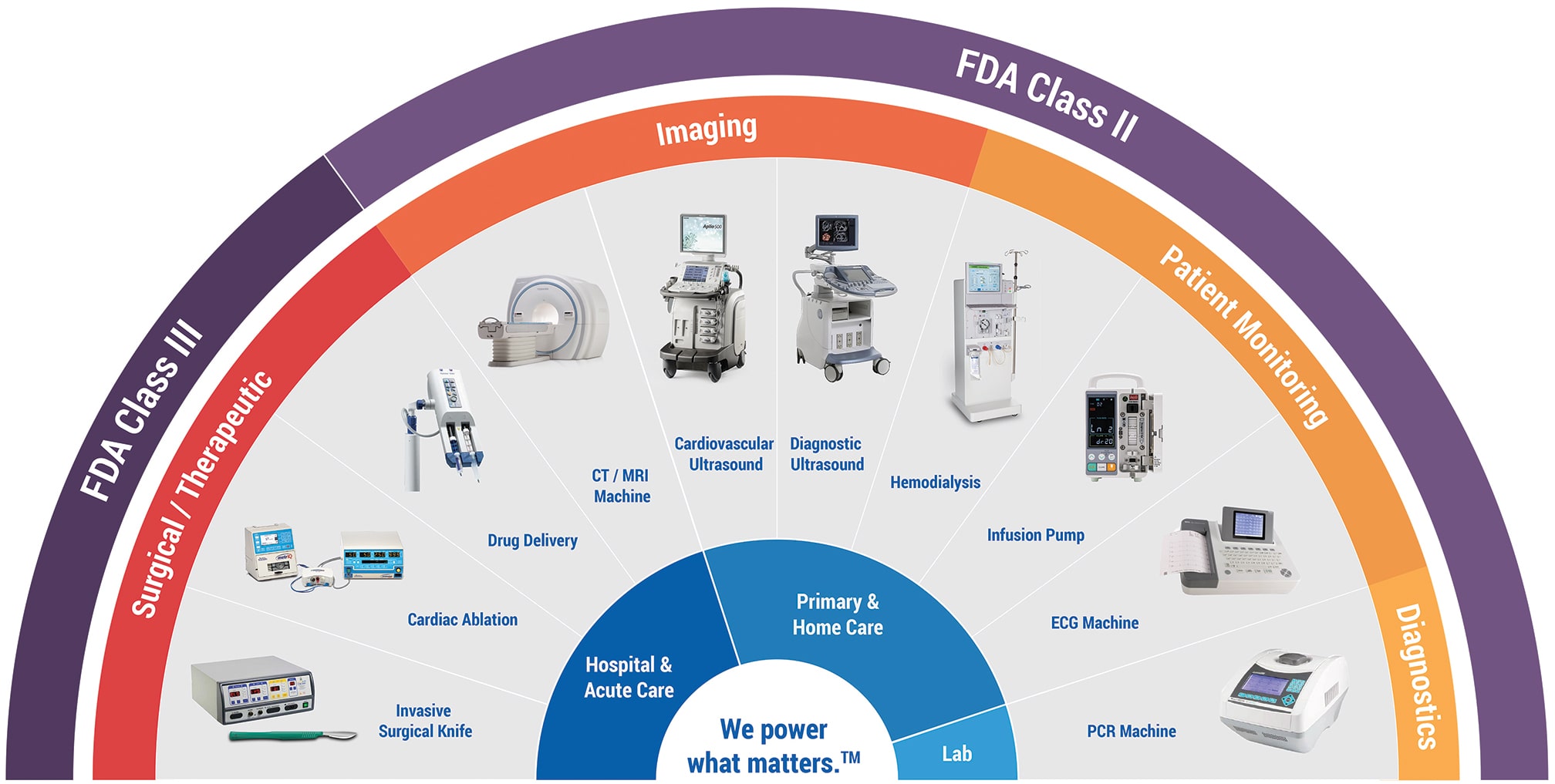
SL Power medical power supplies can be found in virtually every healthcare application category, including surgical technologies, patient monitoring and home care applications. In addition to power supplies that meet the Type B classification, we are one of just a handful of companies that offers Type BF and Type CF rated power supplies. These classifications are part of the ‘applied parts’ context of the IEC 60601 specification and relate to parts of a medical device that may come into physical contact with the patient during its normal operation.
Type B (body) is the least stringent classification, covering parts which are not normally conductive and can be immediately released from the patient. Examples would be operating theatre lighting, medical lasers, MRI scanners, hospital beds and phototherapy equipment.
Type BF (body floating) covers equipment that comes into conductive contact with the patient, whether for a short or long time. It is probably the largest category of medical equipment and includes devices such as blood pressure monitors, non-cardiac surgical equipment, incubators and ultrasound equipment.
The most stringent classification, Type CF (cardiac floating), is for applied parts that may come into direct contact with the patient’s heart. A Type CF power supply provides a much higher degree of protection against electric shock and much tighter leakage current limits to the patient than a Type BF unit. This category includes medical devices for dialysis or ablation and electrocardiograph (ECG) machines.
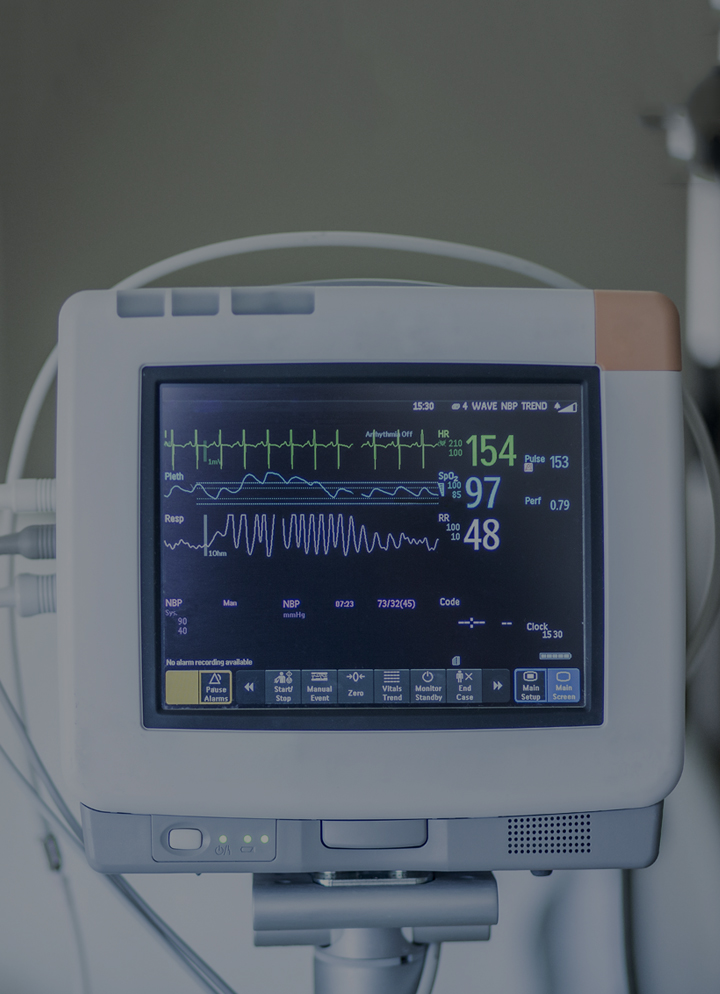
Power supplies for patient monitoring equipment may be external adapters or internal embedded units. The power supply may be powering sensors for pulse oximetry, blood pressure or body temperature, a mechanism for inflating a cuff, valves or pumps, in addition to the internal circuitry and possibly a touch screen controller. As patient monitoring systems become more mobile, battery charging and communications modules have become a critical part of the draw on a patient monitoring power supply.
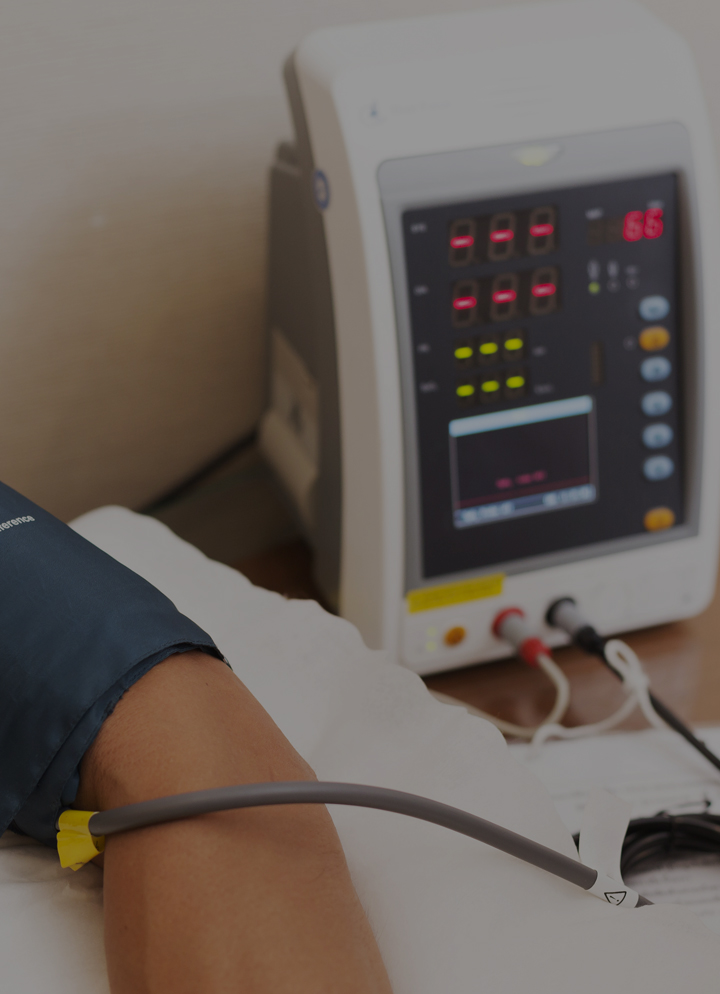
Home healthcare equipment includes devices such as a CPAP or infusion pump. This medical equipment typically uses an external adapter and, given the environment, usually a Class II input unit. Class II input means that power supply requires no earth ground connection, which makes it ideal for use in applications where it is more likely to come into contact with a member of the public. IEC protection Class II power supplies have a two-wire power cord rather than a three-wire power cord with a safety earth connection.
If you would like more information, please click on the button below.